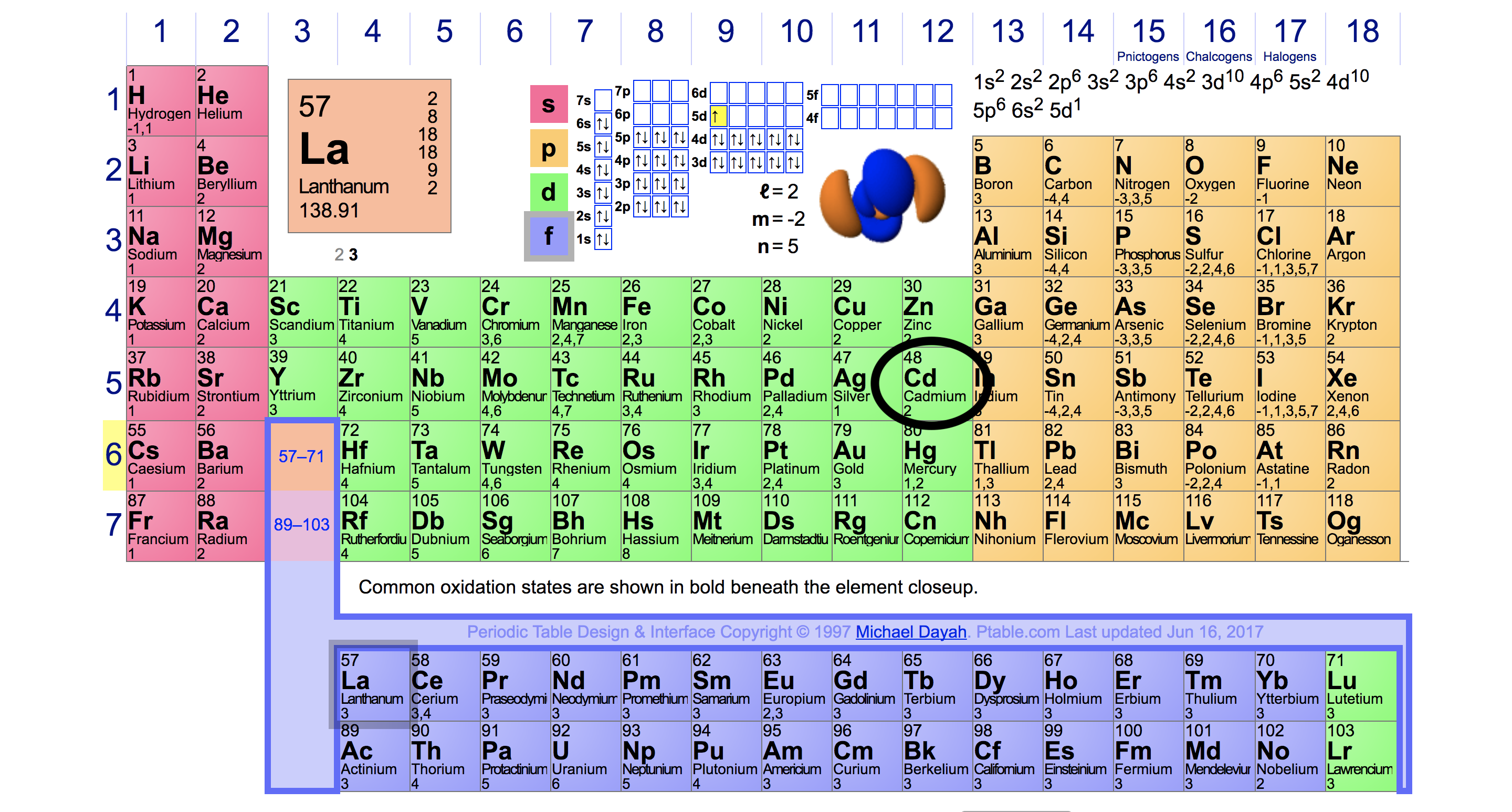
Point to Remember: Mostly for transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core. (See the above or below periodic table to see the valence electrons of inner transition metals). Inner transition elements can have valence electrons ranging from 3 to 16. In some inner transition metals, the electrons of incomplete d-orbitals are also considered as valence electrons. Figure 2 2: Within each period, the trend in atomic radius decreases as Z increases for example, from K to Kr. Typically, you need at least 8 steps to determine the electron configuration, starting with finding the atomic number by looking at the list of orbitals and understanding the notation. The stronger pull (higher effective nuclear charge) experienced by electrons on the right side of the periodic table draws them closer to the nucleus, making the covalent radii smaller. Hence, for inner transition elements, the electrons of both f-subshells as well as s-subshell behave like valence electrons. This electron configuration calculator will instantly show you the distribution of electrons in the orbitals of any periodic element you choose. The inner transition elements have incomplete f- subshells and they are very close to the outer s-subshell. (See the above or below periodic table to see the valence electrons of transition metals).Īlso the two bottom rows at the bottom of the periodic table are the inner transition elements (or f-block elements) also have the similar case. Hence, the transition elements (i.e d-block elements from group 3 to 12) can have more valence electrons ranging from 3 to 12. Draw an orbital diagram and use it to derive the electron configuration of chlorine, Z 17. This gives a valence-electron configuration of 3 s2 3 p3. So, the electrons of both d-subshell and s-subshell behave like valence electrons. C We obtain the valence electron configuration by ignoring the inner orbitals, which for phosphorus means that we ignore the Ne closed shell. It is more difficult to find the valence electrons of transition elements as they have incompletely filled d-subshell and this d-subshell is very close to the outer s-subshell. What about valence electrons of transition and inner transition elements?įor the transition elements and inner transition elements, the case is more complicated.
.jpg)
Well, this suits perfectly for the main group elements (i.e group 1, 2 and group 13 to 18), but what about the transition and inner transition elements? Hence, magnesium has 2 valence electrons. Here, you can see that the highest principal quantum number is 3, and the total electrons in this principal quantum number is 2. The electron configuration of magnesium is 1s 2 2s 2 2p 6 3s 2. Group 3-12: Transition and Inner transition metals group.
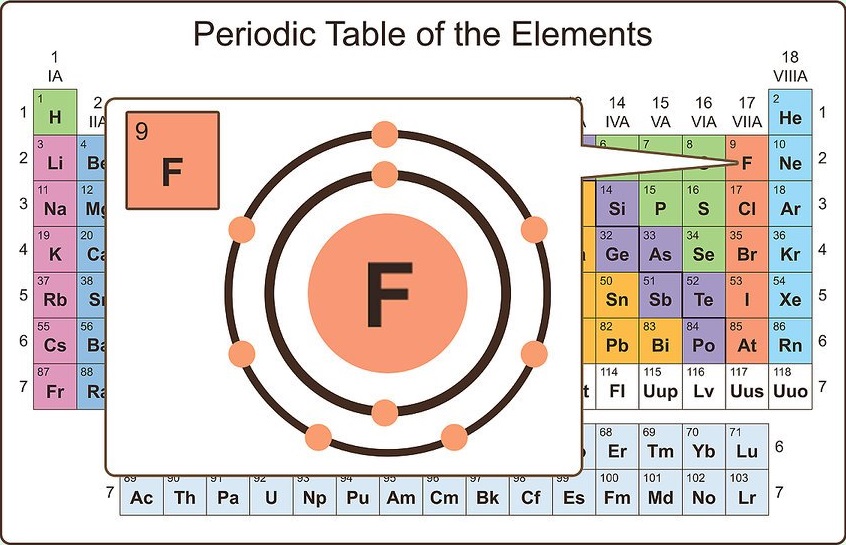
Group 1: Alkali metals group (hydrogen not included) Group 2: Alkaline earth metals group. Valence electrons can also be determined as the electrons present in the shell with highest principal quantum number (n). There are total 18 different groups in Periodic table. The magnesium element has 2 electrons in outermost orbit.
